
Introduction
High-throughput labs processing hundreds of samples daily face a shared bottleneck: DNA extraction. When extraction capacity falls behind sample volume, turnaround times slip and downstream testing queues back up fast.
In oncology diagnostics, large-scale sequencing facilities, and clinical testing centers, the pressure is acute. Labs running tumor profiling panels, prenatal screens, or infectious disease surveillance cannot afford workflows that demand constant batching, manual intervention, or rigid throughput ceilings.
Not all automated DNA extraction kits solve this equally. Throughput capacity, extraction chemistry, open versus closed system architecture, and regulatory certification status all affect result quality and lab economics.
A kit built for biobank-scale batch processing may fail a clinical lab that needs rapid on-demand extractions. Conversely, a compact system suited to variable loads may lack the validation required for IVD diagnostics.
This guide compares five leading automated DNA extraction kits built for high-throughput labs, covering what sets them apart in chemistry, throughput design, regulatory status, and application fit—so you can match the right platform to your workflow.
TL;DR
- Magnetic bead chemistries dominate high-throughput DNA extraction due to speed, scalability, and run-to-run reproducibility
- Open systems offer reagent flexibility; closed systems provide vendor-optimized workflows suited for regulatory compliance
- Key evaluation criteria: samples per run, hands-on time, open vs. closed design, NGS/PCR/long-read compatibility, and IVD certifications
- The five kits compared here range from compact clinical extractors to large-scale biobank platforms across multiple throughput tiers
Automated DNA Extraction in High-Throughput Labs
Automated DNA extraction refers to robotic or semi-automated systems that move samples through lysis, binding, washing, and elution without manual pipetting. These platforms enable labs to process large batches consistently, reducing operator variability and freeing skilled staff from repetitive tasks.
The global nucleic acid isolation and purification market was valued at $3.02 billion in 2025 and is projected to grow at 9.8% CAGR, driven by molecular diagnostics, genomic research, and precision medicine. Across these fields, the extraction kit you choose directly affects cost per test, regulatory compliance, and data quality.
High-growth use cases driving this demand include:
- Oncology labs scaling tumor mutation profiling
- Biobanks archiving large population cohorts
- Infectious disease testing at diagnostic volume
- cfDNA isolation for liquid biopsy workflows
Top Automated DNA Extraction Kits for High-Throughput Labs
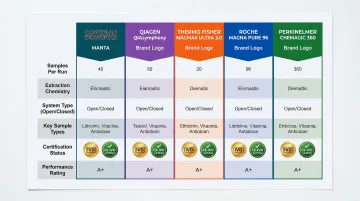
These five kits were selected based on extraction chemistry, throughput range, system compatibility, certification status, and documented performance across clinical and research settings.
Cambrian Bioworks Manta
Manta is a compact, CE-IVD-certified automated nucleic acid extractor designed for clinical and research labs needing flexible, on-demand automation without batching pressure. Trusted by 67+ labs globally including Tata Memorial Hospital and Strand Life Sciences, Manta delivers precision extraction in diverse workflows.
What Sets Manta Apart:
- Uses patented CamSelect Long™ magnetic bead technology enabling tunable size selection for high-molecular-weight DNA enrichment
- Completes standard DNA extraction in ~30 minutes
- Compact footprint (43.5 cm × 28.2 cm × 26.2 cm) fits inside biosafety hoods
- Open-system compatibility with third-party magnetic bead kits
- UVC lamp for contamination control plus touchscreen interface with remote monitoring
- Elutes into barcoded tubes for traceability
| Feature | Details |
|---|---|
| Throughput & Run Flexibility | 1–32 samples per run; no batching pressure; true random-access processing |
| Extraction Technology & Sample Types | Magnetic bead-based; blood, tissue, FFPE, cfDNA, saliva, swabs, stool; ~30 min standard extraction |
| Certifications & Regulatory Status | CE-IVD (IVDR), CDSCO IVD, ISO 13485; available in 5+ countries |

Qiagen QIAsymphony DSP DNA Kit
The QIAsymphony platform is a widely adopted closed-system automated extractor used in clinical and reference labs globally. Its DSP (Diagnostic Sample Preparation) kits are validated for IVD use with extensive regulatory clearances.
Key Differentiators:
- High throughput capacity up to 96 samples per run
- Continuous random-access loading allows flexible sample intake
- Walk-away automation with on-board reagent management and LIMS connectivity
- Prefilled reagent cartridges minimize setup time
- Delivers A260/A280 ratios of 1.7–1.9 consistently
| Feature | Details |
|---|---|
| Throughput & Run Flexibility | Up to 96 samples per run; continuous random-access loading |
| Extraction Technology & Sample Types | Magnetic particle technology; blood, swabs, tissue, CSF; variable elution volumes |
| Certifications & Regulatory Status | IVD-cleared; CE-IVD; validated protocols for clinical diagnostics |
Thermo Fisher Applied Biosystems MagMAX DNA Multi-Sample Ultra 2.0 Kit
The MagMAX DNA Multi-Sample Ultra 2.0 Kit is engineered for high-volume genomics workflows, particularly NGS and long-read sequencing applications, compatible with KingFisher instrument platforms.
Key Differentiators:
- Optimized for high-quality genomic DNA suitable for demanding sequencing workflows
- Scalable bulk reagent formats for high-volume labs
- 20–30 minutes hands-on time with automated protocol support
- Compatible with KingFisher Duo Prime and Flex platforms
- Delivers consistent purity with A260/280 ratios of 1.8–2.0
| Feature | Details |
|---|---|
| Throughput & Run Flexibility | Scalable from small to large runs; bulk formats; 96-well processing |
| Extraction Technology & Sample Types | Magnetic bead-based; blood, tissue, cultured cells; optimized for genomic DNA quality |
| Certifications & Regulatory Status | Research use only; instrument-dependent validation; no standalone IVD claim |
Roche MagNA Pure 96 DNA and Viral NA Small Volume Kit
The Roche MagNA Pure 96 is a fully automated, closed-system extractor built for medium-to-high-throughput clinical labs. It targets infectious disease, oncology, and virology panels that demand high DNA and viral nucleic acid purity.
Key Differentiators:
- Walk-away automation for up to 96 samples per run
- Magnetic glass particle technology ensures high purity
- Prefilled reagent cartridges and barcode scanning
- LIMS integration for connected lab workflow
- FDA-registered and CE-IVD certified
- Less than 5 minutes hands-on time per run
| Feature | Details |
|---|---|
| Throughput & Run Flexibility | 96 samples per run; fully automated with barcode scanning and LIMS connectivity |
| Extraction Technology & Sample Types | Magnetic glass particle technology; blood, plasma, swabs, tissue, urine; dual DNA/RNA extraction available |
| Certifications & Regulatory Status | CE-IVD; FDA-registered; IVD-validated protocols |
PerkinElmer Chemagic 360 DNA Kit
The Chemagic 360 by PerkinElmer (now Revvity) is a high-capacity automated platform designed for biobanking, large-scale epidemiological studies, and high-volume reference labs. It handles large sample batches reliably, making it a common choice for population-scale genomics programs.
Key Differentiators:
- Processes up to 96 samples per run with 96-rod head configuration
- M-PVA magnetic bead chemistry optimized for high yields
- Supports large input volumes from 50 µL up to 18 mL
- Highly suitable for saliva, blood, and tissue samples
- Designed for continuous high-volume biobank-scale operations
| Feature | Details |
|---|---|
| Throughput & Run Flexibility | Up to 96 samples per run with 96-rod head; large input volume support (50 µL–18 mL) |
| Extraction Technology & Sample Types | M-PVA magnetic bead technology; blood, saliva, tissue, cells; high-yield genomic DNA |
| Certifications & Regulatory Status | Research and diagnostic use; CE-marked instrument; kit IVD status varies by region |
Key Criteria for Comparing Automated DNA Extraction Kits
Throughput and Run Flexibility
Fixed-batch systems require a full plate (typically 96 samples) to run efficiently. That works well for biobanks processing daily cohorts — but creates real bottlenecks in oncology or prenatal labs where sample volumes are unpredictable.
A clinic receiving 15 urgent tumor samples cannot hold extraction until 96 samples accumulate. The delay defeats the purpose of rapid diagnostics.
Flexible systems allow variable sample numbers per run—processing anywhere from one to full capacity without wasting consumables or holding up turnaround. Labs handling sporadic sample intake benefit significantly from this architecture.
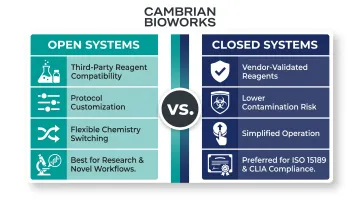
Open vs. Closed System Architecture
The choice between open and closed architecture shapes how much control — and how much compliance overhead — your lab takes on:
- Open systems (liquid handlers compatible with third-party reagents) allow protocol customization, chemistry switching across sample types, and flexibility as new assays emerge. Best fit for research labs or diagnostics teams validating novel workflows.
- Closed systems run vendor-specified protocols with pre-validated reagents, offering simpler operation, lower contamination risk, and the regulatory documentation required for clinical accreditation.
For labs operating under ISO 15189 or CLIA, closed systems significantly reduce validation burden and audit risk.
DNA Quality and Downstream Application Fit
Labs running NGS or long-read sequencing need high-molecular-weight DNA with fragment sizes exceeding 10 kb for optimal read lengths. PacBio recommends 50–100 kb HMW DNA, while Illumina requires A260/280 ratios of 1.8–2.0 for library preparation.
PCR-based clinical diagnostics can tolerate shorter fragments, but elution volume, yield, and purity metrics must still align with downstream instrument input requirements. A kit delivering 50 µL elution may require concentration steps before loading into assays needing higher DNA mass.
Certifications and Regulatory Compliance
For clinical diagnostic labs, IVD certification is non-negotiable. CE-IVD under IVDR 2017/746, CDSCO clearance in India, or FDA 510(k) clearance in the US each indicate that the kit meets validated performance standards for diagnostic use. Choosing a research-use-only kit in an accredited environment creates direct compliance risk and can void lab certifications.
When reviewing manufacturer credentials, ISO 13485 certification confirms that quality management systems meet medical device production standards — a relevant factor for procurement and supplier qualification audits.
Total Cost of Ownership Beyond Kit Price
Labs often underestimate costs by focusing only on reagent list price. Total cost of ownership includes:
- Instrument capital cost and depreciation
- Maintenance contracts and service calls
- Consumable waste (tips, plates, cartridges)
- Cost per test including labor
- Space and infrastructure requirements
In a customer workflow analysis, labs using Manta reported a 56% reduction in cost per test — driven by 24% less plastic waste, 74% faster turnaround, and the elimination of batching delays. When evaluating platforms, calculate cost per test against your actual sample volume, not theoretical maximum throughput.

How We Chose These Kits
These kits were evaluated on criteria that reflect real workflow demands — not brand reputation alone:
- Extraction chemistry type and underlying mechanism
- Validated throughput range across sample batches
- Sample type compatibility (blood, FFPE, swabs, cfDNA, etc.)
- Open vs. closed system design and instrument lock-in implications
- Regulatory certification status (CE-IVD, CDSCO, RUO)
- Documented use in clinical or high-throughput research settings
These criteria exist because the most common selection mistakes aren't about quality — they're about fit. Labs routinely choose kits that misalign with their actual workload or downstream requirements:
- Throughput mismatch: A biobank-scale system handling 360 samples per run adds no value to a clinic processing 20 samples daily.
- Regulatory incompatibility: A research-use (RUO) kit may deliver excellent yields but creates compliance gaps in diagnostic settings.
- Brand over workflow: Selecting based on name recognition without assessing protocol fit leads to costly mid-run adaptations.
Conclusion
The right automated DNA extraction kit depends on precise alignment between throughput needs, sample types, downstream applications, and regulatory requirements. A compact, flexible system fits clinical diagnostics with variable volumes; large-batch platforms suit biobanking and epidemiological studies. The best choice is the one that matches your lab's actual operating conditions.
Before committing, run through this evaluation checklist:
- Request demo runs using your actual sample types, not vendor-supplied reference samples
- Evaluate cost per test, not just upfront kit price
- Ask vendors for validation data specific to your sample matrix and downstream application
- Confirm regulatory certifications match your operational environment
- Verify throughput architecture fits your daily workflow
If your lab needs a compact, CE-IVD-certified, magnetic bead-based system — one that handles variable sample loads without batching pressure and works with open-system configurations — Cambrian Bioworks' Manta is designed for exactly that use case. Contact connect@cambrianbioworks.com to explore kit compatibility and arrange a demo with your sample types.
Frequently Asked Questions
What are the different types of DNA isolation kits?
The three main types are magnetic bead-based kits (most common in automation due to speed and reproducibility), spin-column kits (widely used in manual workflows), and precipitation-based kits (employed in some high-yield applications). Magnetic bead systems are preferred for automated high-throughput settings because they eliminate centrifugation steps and enable robotic handling.
How can DNA extraction be optimized for high yield?
Key levers include matching lysis conditions to your sample type, selecting bead chemistry for the required fragment size, minimizing freeze-thaw cycles, and calibrating elution volume to your downstream application. Larger elution volumes increase total yield but reduce concentration; smaller volumes do the reverse.
What is the difference between open and closed automated DNA extraction systems?
Open systems (such as Tecan or Hamilton liquid handlers) accept any extraction chemistry and suit research labs running diverse applications. Closed systems (such as QIAsymphony or MagNA Pure) use vendor-specified, pre-validated reagents — preferred in regulated clinical settings where cross-contamination risk and validation burden must be controlled.
What sample types are compatible with automated DNA extraction kits?
Most automated kits support blood (EDTA and dried blood spots), swabs, tissue (fresh and FFPE), saliva, urine, and plasma. Always verify compatibility against the kit's validated protocol — some chemistries struggle with challenging matrices like stool or low-input cfDNA samples.
What certifications should I look for in a clinical-grade DNA extraction kit?
Clinical labs should prioritize CE-IVD certification under EU IVDR 2017/746 (required for Europe and UK), CDSCO IVD certification for labs in India, and ISO 13485 quality management compliance from the manufacturer. These confirm the kit meets validated diagnostic performance standards and fits into accredited lab workflows.
How many samples can a typical automated DNA extraction system process per run?
Throughput scales by platform: compact clinical systems handle 1–32 samples per run, mid-range platforms manage 48–96, and biobank-scale systems process 96+. Select based on average daily volume rather than peak capacity — oversizing a system leads to underutilization, while undersizing creates batching delays.


