
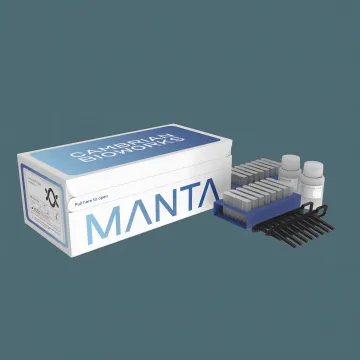
Cambrian Whole Blood gDNA Isolation Kit
- Under 30-min total processing time
- A260/280 ratio 1.8–2.0 for pure gDNA
- Compatible with blood, cultures & bacteria
ISO 13485 Certified
CE-IVD Mark
CDSCO IVD Certified
Cambrian Bioworks' molecular diagnostic extraction kits deliver certified, high-quality nucleic acid isolation from FFPE tissue, whole blood, and plasma. Leveraging magnetic bead-based technology, these ISO 13485 and CE-IVD certified kits support NGS, PCR, liquid biopsy, and oncology workflows with fast, automated, reproducible results.

Talk to our product experts for custom solutions.
Apply Filters





The primary quality certificate for medical devices is ISO 13485, an internationally recognized standard for quality management systems in medical device design and manufacturing. Additionally, devices sold in the EU require CE-IVD marking under IVDR (EU) 2017/746. In India, CDSCO IVD certification is required. Cambrian Bioworks holds all three, ensuring its extraction kits meet rigorous global quality and safety standards.
Talk to our experts for custom solutions and tailored guidance.
Extraction kits power NGS, liquid biopsy, MRD detection, and tumor profiling workflows in cancer diagnostic labs.

Automated, CE-IVD certified extraction kits enable reliable gDNA and RNA isolation for clinical molecular testing.

High-purity nucleic acid kits support NGS library preparation, transcriptomics, and biomarker discovery in sequencing labs.

Whole blood and plasma extraction kits enable sensitive nucleic acid isolation for infectious disease detection and monitoring.
Talk to our experts for custom solutions and tailored guidance.
Every extraction kit is designed and manufactured under ISO 13485 quality management and CE-IVD certification, ensuring clinical-grade reliability and regulatory compliance for diagnostic labs.
From Tata Memorial Hospital to leading sequencing labs globally, Cambrian Bioworks is a proven partner for oncology, NGS, and clinical molecular diagnostic workflows.
CamSelect Long™ Bead Technology and pre-filled cartridge automation deliver consistent high-molecular-weight nucleic acid yields, cutting cost per test by up to 56% and turnaround time by 74%.
Submit your inquiry and our team will respond within one business day with pricing, tester kit options, and demo scheduling.
For immediate assistance, feel free to give us a direct call at +91 78922 39793. You can also send us a quick email at connect@cambrianbioworks.com.
For immediate assistance, feel free to give us a direct call at +91 78922 39793. You can also send us a quick email at connect@cambrianbioworks.com.