
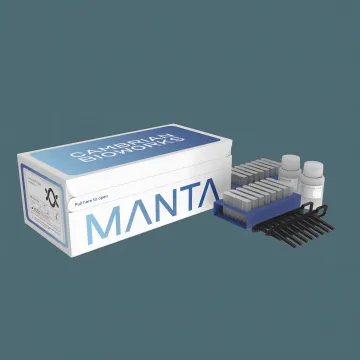
Cambrian Whole Blood gDNA Isolation Kit
- Complete gDNA extraction in under 30 min
- A260/280 ratio 1.8–2.0, NGS-grade purity
- Flexible 200 µL–2 mL whole blood input
ISO 13485 Certified
CE-IVD Mark
CDSCO IVD Certified
Cambrian Bioworks offers CDSCO-approved nucleic acid extraction and molecular diagnostic devices, including kits for FFPE tissue gDNA, whole blood gDNA, and whole blood RNA isolation. Powered by magnetic bead technology on the automated Manta platform, these kits deliver fast, reproducible results for NGS, PCR, and clinical diagnostics.




CDSCO classifies medical devices into four risk-based categories: Class A (low risk), Class B (low-moderate risk), Class C (moderate-high risk), and Class D (high risk). In vitro diagnostic devices like nucleic acid extraction kits are regulated under Schedule II of the Medical Devices Rules, 2017, with classification based on intended use and patient risk.
Talk to our experts for custom solutions and tailored guidance.

FFPE tissue and blood-based DNA/RNA extraction for NGS, mutation analysis, and tumor biomarker profiling.

Automated whole blood gDNA and RNA extraction for genetic testing, disease diagnostics, and variant interpretation.

Rapid nucleic acid isolation from whole blood for PCR-based pathogen detection and infectious disease diagnostics.

High-quality gDNA and RNA extraction from diverse samples supporting NGS, RNA-Seq, and transcriptomics research workflows.
Talk to our experts for custom solutions and tailored guidance.
CDSCO IVD, CE-IVD, and ISO 13485 certifications ensure every kit meets rigorous quality and safety standards for clinical diagnostics.
Automated Manta platform cuts extraction time to under 30 minutes and reduces cost per test by up to 56%, boosting lab productivity.
From Tata Memorial Hospital to leading sequencing labs, Cambrian Bioworks is the trusted extraction partner for research and diagnostics.
Submit your query and our team will respond within one business day with pricing, demo scheduling, and technical guidance.
For immediate assistance, feel free to give us a direct call at +91 78922 39793. You can also send us a quick email at connect@cambrianbioworks.com.
For immediate assistance, feel free to give us a direct call at +91 78922 39793. You can also send us a quick email at connect@cambrianbioworks.com.